Eggs are important for healing because they contain 9 essential amino acids, most athletes need it for muscle building and my dad used to drink raw eggs to heal his TB (lungs). Histidine () and L…
Source: Raw eggs for healing
Affordable in home care | starts at $28 per hr
Eggs are important for healing because they contain 9 essential amino acids, most athletes need it for muscle building and my dad used to drink raw eggs to heal his TB (lungs). Histidine () and L…
Source: Raw eggs for healing
Eggs are important for healing because they contain 9 essential amino acids, most athletes need it for muscle building and my dad used to drink raw eggs to heal his TB (lungs). Histidine () and Leucine are two of the 9 essential amino acids in eggs. Eggs plus Vitamin B rich foods are happy foods, helping happy hormones in the brain.
Eggs contain Histidine which is an amino acid that is used to develop and maintain healthy tissues in all parts of the body, particularly the myelin sheaths that coat nerve cells and ensure the transmission of messages from the brain to various parts of the body. It may be useful for treatment of mental disorders as well as certain types of sexual dysfunction. Histidine levels in the body must be balanced to ensure good mental and physical health. High levels of this amino acid have been linked to the presence of psychological disorders such as anxiety and schizophrenia, while low levels of histidine are thought contribute to the development of rheumatoid arthritis and the type of deafness that results from nerve damage. Taking histidine supplements may help relieve symptoms of rheumatoid arthritis.
Histidine in eggs is important to normal sexual functioning, because it gets converted into histamine, a chemical needed to stimulate sexual arousal. When taken together with vitamin B3 (niacin) and vitamin B6 (pyridoxine), histidine can increase sexual pleasure by boosting histamine levels in the body. Histamine is also needed to help the immune system know when the body is experiencing an allergic reaction, and for the production of gastric juices needed for normal digestion. Research suggests that hsitidine also acts as a natural detoxifier, protecting against radiation damage, and removing heavy metals from the system. It may even help prevent the onset of AIDS—histidine is crucial to the production of both red and white blood cells. Like other amino acids, histidine is found in many high-protein foods such as raw eggs.
Leucine works with the amino acids isoleucine and valine to repair muscles, regulate blood sugar, and provide the body with energy. It also increases production of growth hormones, and helps burn visceral fat, which is located in the deepest layers of the body and the least responsive to dieting and exercise.
Leucine, isoleucine, and valine are branched-chain amino acids (BCAAs), and all three of them help promote muscle recovery after exercise. Leucine is the most effective BCAA for preventing muscle loss because it breaks down and is converted to glucose more quickly than isoleucine and valine. Increased glucose supplies prevent the body’s cannibalization of muscle for energy during intense workouts, so it is no surprise that this amino acid supplement is popular among professional body builders. Leucine also promotes the healing of bones, skin, and muscle tissue after traumatic injury, and is often recommended for those recovering from surgery. Because it is so easily converted to glucose, leucine helps to regulate blood sugar; a deficiency of leucine produces symptoms similar to those of hypoglycemia, which may include headaches, dizziness, fatigue, depression, confusion, and irritability.
Histamine is also essential in defending the body against invasion by potentially disease-causing agents such as bacteria, viruses and other foreign invaders. Histamine is made and stored within white blood cells (leukocytes) such as mast cells in tissues and basophils that circulate in blood. When the immune system is activated in response to foreign material entering the body, histamine is the first “defence chemical”, or more correctly, inflammatory mediator, released in the process called inflammation.
Foods with High Histamine levels: Long cooked or leftover meat, Most Alcohol (Wine, Beer, Cider, Fermented Drinks), Yeast, Fish that is not immediately gutted after catching, Aged Fish (Canned, Smoked), Tomatos (Fresh or Processed), Pickled, Fermented & Cultured Foods Sauerkraut Pickles, Kimchee, Soy Sauce, Tamari, Miso), Smoked & Cured Meats (Ham, Sausage, Salami), Shell Fish, Red Beans, Nuts, Chocolate, Citrus Fruit, Wheat Based Foods, Vinegar (Salad Dressings, Mustard, Ketchup, Mayonnaise), Spices & Seasonings (Cinnamon, Nutmeg, Hot Peppers, Cloves, Anise, Curry, Chili Powder, Overripe Fruits, Yogurt & Kefir (depends upon the cultures used), Canned Food ( additives & preservatives), Soy Products, Mushrooms, Dried Fruits, Cola, Raw & Undercooked Egg (Whites in particular)
My grandma would burn the egg yolk to a black paste and apply it on my feet with eczema/athlete’s foot and it healed my skin (Sulfur is the burnt black chemical).
Dr Bernard Jensen wrote in his book, The Chemistry of Man, that yellow rich foods are also rich in cleansing to the body compound, Sulfur.
Dr Ken Giuffre wrote: Frequently overlooked essential signs and symptoms when screening for Alzheimer, Parkinson’s and Anxiety Disorder: Depression and anxiety, or any new other new mood chan…
Source: Signs and symptoms when screening for Alzheimer, Parkinson’s and Anxiety Disorder
Dr Ken Giuffre wrote:
Frequently overlooked essential signs and symptoms when screening for Alzheimer, Parkinson’s and Anxiety Disorder:
Connie’s comments:
Slowness in speech, senior moments, gait issues, penmanship issues, lack of taste and your taste when kissing your loved ones, changes in blood and urine tests results, and many other subtle signs that aging happens or some health issues should be checked by the doctor early on.
Restless leg syndrome, cramps and migraines, diabetes and high blood pressure are also important indicators if our body is needing more health care needs.
What our family eats when we are growing up can impact our health and also what our mothers ate when we are conceived and growing in her womb. Breastfeeding and the age and lifestyle (alcohol,smoking) of our fathers can have a greater impact to our health.
Western diet and lifestyle mold our intestinal microbiome that those in farming focus non industrialized countries have more healthy babies.
It is not too late, move/exercise, sing an dance and eat only whole foods and only when hungry.
Yale scientists have solved a puzzle of the immune system—how antibodies enter the nervous system to control viral infections. Their finding may have implications for the prevention and treatment of a range of conditions, including herpes and Guillain-Barre syndrome, which has been linked to the Zika virus.
Many viruses, such as West Nile, Zika, and the herpes simplex virus enter the nervous system, where they were thought to be beyond the reach of antibodies. Yale immunobiologists Dr. Akiko Iwasaki and Norifumi Iijima used mice models to investigate how antibodies could gain access to nerve tissue in order to control infection.
In mice infected with herpes, they observed a previously under-recognized role of CD4 T cells, a type of white blood cell that guards against infection by sending signals to activate immunity.
“This is a very elegant design of the immune system to allow antibodies to go to the sites of infection,” said Iwasaki. “The CD4 T cells will only go to the site where there is a virus. It’s a targeted delivery system for antibodies.”
The implications of the finding are multiple. Without CD4 T cells, antibody-based therapies that are being developed for conditions like herpes may not be sufficient to control infection, Iwasaki noted. Conversely, for antibody-mediated autoimmune diseases such as Guillain-Barre, “it may be beneficial to block CD4 from entering the neuronal tissues,” she said.
Explore further: New vaccine strategy may fight genital herpes, mouse study suggests
More information: Norifumi Iijima et al, Access of protective antiviral antibody to neuronal tissues requires CD4 T-cell help, Nature (2016). DOI: 10.1038/nature17979
Journal reference: Nature search and more info website
Provided by: Yale University
Eur J Immunol. 1990 Aug;20(8):1877-80.
Bertotto A1, Gerli R, Fabietti G, Crupi S, Arcangeli C, Scalise F, Vaccaro R.
Naive (unsensitized) and memory (antigen-primed) T cells can be phenotypically distinguished on the basis of the high or low intensity with which they express a number of immunologically relevant lymphocyte membrane antigens, including CD45R, CDw29, UCHL1, LFA-1, LFA-3, CD2 and Pgp-1. Here we report that in contrast to the two major T cell subsets found in the blood, milk T lymphocytes are almost exclusively composed of the one which exhibits the CD45Rlow, CDw29, UCHL1, LFA-1high memory T cell phenotype. In addition, while milk and autologous blood cells expressed similar levels of CD3 surface antigens, CD2 and ICAM-1 expression was approximately twofold greater on the milk T lymphocytes. This agrees with the finding that whereas colostrum T cells respond poorly to PHA, they proliferate and produce interferon-gamma normally when stimulated with either the anti-CD3 or anti-CD2 monoclonal antibodies. The selective colonization of the mammary gland during lactation by a population of T lymphocytes which displays the phenotype and functional characteristics of memory T cells may be one of the mechanisms whereby the suckling infant benefits from its mother’s immunological experience.
Antibodies, which are also called immunoglobulins, take five basic forms, denoted as IgG, IgA, IgM, IgD and IgE. All have been found in human milk, but by far the most abundant type is IgA, specifically the form known as secretory IgA, which is found in great amounts throughout the gut and respiratory system of adults. These antibodies consist of two joined IgA molecules and a so-called secretory component that seems to shield the antibody molecules from being degraded by the gastric acid and digestive enzymes in the stomach and intestines. Infants who are bottle-fed have few means for battling ingested pathogens until they begin making secretory IgA on their own, often several weeks or even months after birth.
The secretory IgA molecules passed to the suckling child are helpful in ways that go beyond their ability to bind to microorganisms and keep them away from the body’s tissues. First, the collection of antibodies transmitted to an infant is highly targeted against pathogens in that child’s immediate surroundings. The mother synthesizes antibodies when she ingests, inhales or otherwise comes in contact with a disease-causing agent.
The molecules in milk have other valuable functions as well. Each molecule of a protein called lactoferrin, for example, can bind to two atoms of iron. Because many pathogenic bacteria thrive on iron, lactoferrin halts their spread by making iron unavailable. It is especially effective at stalling the proliferation of organisms that often cause serious illness in infants, including Staphylococcus aureus. Lactoferrin also disrupts the process by which bacteria digest carbohydrates, further limiting their growth. Similarly, B12 binding protein, as its name suggests, deprives microorganisms of vitamin B12. Bifidus factor, one of the oldest known disease-resistance factors in human milk, promotes the growth of a beneficial organism named Lactobacillus bifidus. Free fatty acids present in milk can damage the membranes of enveloped viruses, such as the chicken pox virus, which are packets of genetic material encased in protein shells. Interferon, found particularly in colostrum-the scant, sometimes yellowish milk a mother produces during the first few days after birth-also has strong antiviral activity. And fibronectin, present in large quantities in colostrum, can make certain phagocytes more aggressive so that they will ingest microbes even when the microbes have not been tagged by an antibody. Like secretory IgA, fibronectin minimizes inflammation; it also seems to aid in repairing tissue damaged by inflammation.
The next most common milk leukocyte is the macrophage, which is phagocytic like neutrophils and performs a number of other protective functions. Macrophages make up some 40 percent of all the leukocytes in colostrum. They are far more active than milk neutrophils, and recent experiments suggest that they are more motile than are their counterparts in blood. Aside from being phagocytic, the macrophages in breast milk manufacture lysozyme, increasing its amount in the infant’s gastrointestinal tract. Lysozyme is an enzyme that destroys bacteria by disrupting their cell walls.
In addition, macrophages in the digestive tract can rally lymphocytes into action against invaders. Lymphocytes constitute the remaining 10 percent of white cells in the milk. About 20 percent of these cells are B lymphocytes, which give rise to antibodies; the rest are T lymphocytes, which kill infected cells directly or send out chemical messages that mobilize still other components of the immune system. Milk lymphocytes seem to behave differently from blood lymphocytes. Those in milk, for example, proliferate in the presence of Escherichia coli, a bacterium that can cause life-threatening illness in babies, but they are far less responsive than blood lymphocytes to agents posing less threat to infants. Milk lymphocytes also manufacture several chemicals-including gamma-interferon, migration inhibition factor and monocyte chemotactic factor-that can strengthen an infant’s own immune response.
It is defect of elastic fiber fragmentation in arterial wall and not hypertension and arterial stiffness that leads to plaque accumulation as we age Medical professionals have long known that the b…
Source: Reduced amounts of elastic fibers as we age causes plaque build-up
Medical professionals have long known that the buildup of plaque in arteries can cause them to narrow and harden, potentially leading to a whole host of health problems—including heart attack, heart disease and stroke. While high blood pressure and artery stiffness are often associated with plaque buildup, new research from engineers at Washington University in St. Louis shows they are not the direct causes.
The team suggests a new culprit: fragmentation of elastic fibers within the arterial wall. The findings, recently published online in the journal Atherosclerosis, could change the scope of heart disease detection and treatment for millions of Americans.
“Our surprising results suggest that treating patients for hypertension and arterial stiffness may have no effect on plaque buildup because we are not treating the underlying defect of elastic fiber fragmentation,” said Jessica Wagenseil, associate professor of Mechanical Engineering & Materials Science in the School of Engineering & Applied Science.
Wagenseil’s lab used two different groups of mice in the study. Some were genetically predisposed to hypertension and reduced aortic compliance, or increased artery stiffness. The other mice were not genetically predisposed to the heart conditions. All of the mice were fed a Western (high-fat) diet for 16 weeks.
Wagenseil’s team hypothesized the mice with the genetic heart issues would have more plaque buildup than the group without. However, their findings proved that hypothesis wrong.
After extensive testing, the team could detect no significant differences in plaque buildup between the two groups of mice. That led them to explore a third factor not typically associated with plaque accumulation: the structure of elastic fibers within the arterial wall.
In aging humans and in previous animal studies, hypertension and increased arterial stiffness are accompanied by fragmentation of the elastic fibers. In Wagenseil’s study, the mice had reduced amounts of elastic fibers, which causes hypertension and increased arterial stiffness, but no elastic fiber fragmentation, which may be the critical difference in plaque accumulation.
“We were able to separate the effects of elastic fiber fragmentation from hypertension and arterial stiffness in plaque accumulation,” Wagenseil said. “We found that hypertension and arterial stiffness alone, without elastic fiber fragmentation, have no effect on plaque buildup.”
In editorial remarks in Atherosclerosis about the research, field experts Laura Hansen and W. Robert Taylor said Wagenseil’s findings “suggest a paradigm shift” in the way researchers think about risk factors for plaque buildup, and the strategies for better treatment options.
The idea that increased artery stiffness is a consequence of plaque buildup, as opposed to a cause of it, as suggested by Wagenseil’s findings, offers a different approach in the study of heart disease. It also could lead to a re-examination of the relationship between increased plaque and diet, exercise and other lifestyle choices commonly associated with artery health.
“Elastic fiber fragmentation is likely a key player in plaque buildup,” Wagenseil said. “The next step is to determine how fragmentation affects the movement and activation of molecules and cells that are involved in plaque formation.”
Explore further: Researchers discover molecule that may prevent atherosclerosis
More information: Justine A. Maedeker et al, Hypertension and decreased aortic compliance due to reduced elastin amounts do not increase atherosclerotic plaque accumulation in Ldlr−/− mice, Atherosclerosis (2016). DOI: 10.1016/j.atherosclerosis.2016.03.022
Provided by: Washington University in St. Louis search and more info website
Connie’s comments:
A molecular pathway that is activated in the brain during fasting helps halt the spread of intestinal bacteria into the bloodstream, according to a new study by a team of researchers at the Salk Institute.
The study, published the week of May 16, 2016 in the Proceedings of the National Academy of Sciences, shows a molecular pathway by which the brain communicates with the gastrointestinal (GI) tract to prevent unnecessary activation of the immune system during fasting by strengthening the barrier against gut microbes. The discovery of this brain-gut signal in fruit flies, which has many parallels to humans, could eventually inform the treatment of inflammatory bowel diseases in people.
In addition to its role in promoting the absorption of nutrients from food, the GI tract is host to a panoply of bacteria. These microbes actually help in the digestive process by producing chemicals that break down complex fats and carbohydrates.
“Fasting has a positive value that spills over not just into the metabolic system, but also inflammation and brain function,” says the study’s lead investigator Marc Montminy, professor in the Clayton Foundation Laboratories for Peptide Biology and holder of the J.W. Kieckhefer Foundation Chair. “Understanding how the gut maintains this barrier, and creating drugs to enhance that barrier, may have important benefits for people with inflammatory bowel disease.”
The new study is part of an ongoing collaborative effort by the Montminy lab and the lab of Salk Professor John Thomas to pin down the mechanisms that a genetic switch in the brain called Crtc uses to control energy balance. A constant network of communication—between our brains and the GI tract, as well as other tissues—helps our bodies keep tabs on our energy expenditure and stores. Crtc interacts with another molecule called CREB, and fasting activates both proteins and boosts formation of long-term memories.
The Montminy and Thomas teams used fruit flies to study the Crtc switch, in part because flies express many of the same metabolism-related genes as humans do. Previous experiments by the two labs have shown that flies whose Crtc gene is deleted become sensitized to fasting—they only survive about half as long without food compared to flies with the Crtc gene. The researchers were aiming to understand why the deletion of Crtc caused flies to die sooner and had hypothesized it was because these mutant flies have fewer fat and sugar stores.
What the team—along with Salk Assistant Professor Janelle Ayres’ group—found in the new study, however, was surprising and more complicated. The guts of the flies without Crtc expressed several molecules indicating that their immune system was keyed up. When postdoctoral researcher Run Shen entered Montminy’s lab with the evidence—pictures taken from the microscope of fluorescently stained cells lining the flies’ guts—”it was totally unexpected,” he says.
The new results suggest that the flies are more sensitive to starvation because the immune system is activated, which is energetically taxing. This amped-up immune response suggests that without Crtc, bacteria leak from the gut into the fly’s circulation. The researchers found that the normal role of Crtc is to fortify the barriers of the gut to prevent bacteria from entering the bloodstream and awakening the immune system. Without Crtc, the connections between cells that line the gut tube become disrupted, causing bacteria to leak out, activating the immune response and depleting energy reserves.
While looking for molecular partners of Crtc, the researchers uncovered a protein called short neuropeptide F (sNPF), which is also found in the brain and has an equivalent in humans (called neuropeptide Y). This peptide is known to cause flies and mammals to search for food in response to hunger signals. Without sNPF in the brain, the flies showed signs of gut inflammation similar to those flies missing Crtc. What’s more, the normally tight seals along the gastrointestinal tract were broken down in the sNPF-lacking flies, letting bacteria out.
Conversely, flies expressing more than the normal amounts of Crtc or sNPF in their neurons were able to survive longer without food and showed less disruption to the tight junctions that maintain their gastrointestinal barriers.
The researchers are conducting more experiments to understand how the neuropeptides activate the gut receptors that help protect it from bacterial invasion.
Explore further: Drowsy Drosophila shed light on sleep and hunger
More information: Neuronal energy-sensing pathway promotes energy balance by modulating disease tolerance, PNAS, http://www.pnas.org/cgi/doi/10.1073/pnas.1606106113
Journal reference: Proceedings of the National Academy of Sciences search and more info website
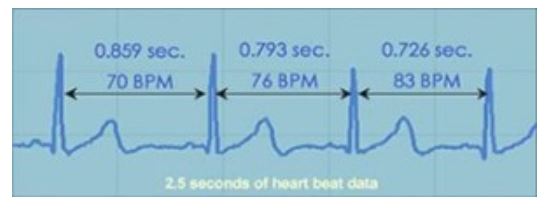
Scientists and physicians consider HRV to be an important indicator of health and fitness. As a marker of physiological resilience and behavioral flexibility, it reflects our ability to adapt effectively to stress and environmental demands. A simple analogy helps to illustrate this point: just as the shifting stance of a tennis player about to receive a serve may facilitate swift adaptation, in healthy individuals the heart remains similarly responsive and resilient, primed and ready to react when needed.
Our heart rate variability is greatest when we are young, and as we age the range of variation in our resting heart rate becomes smaller. Although the age-related decline in HRV is a natural process, having abnormally low HRV for one’s age group is associated with increased risk of future health problems and premature mortality. Low HRV is also observed in individuals with a wide range of diseases and disorders. By reducing stress-induced wear and tear on the nervous system and facilitating the body’s natural regenerative processes, regular practice of HeartMath coherence-building techniques can help restore low HRV to healthy values.
Many factors affect the activity of the ANS, and therefore influence HRV. These include our breathing patterns, physical exercise, and even our thoughts. Research at the Institute of HeartMath has shown that one of the most powerful factors that affect our heart’s changing rhythm is our feelings and emotions. When our varying heart rate is plotted over time, the overall shape of the waveform produced is called the heart rhythm pattern. When you use the emWave and Inner Balance technologies, you are seeing your heart rhythm pattern in real time. HeartMath research has found that the emotions we experience directly affect our heart rhythm pattern – and this, in turn, tells us much about how our body is functioning.
In general, emotional stress – including emotions such as anger, frustration, and anxiety—gives rise to heart rhythm patterns that appear irregular and erratic: the HRV waveform looks like a series of uneven, jagged peaks (an example is shown in the figure below). Scientists call this an incoherent heart rhythm pattern. Physiologically, this pattern indicates that the signals produced by the two branches of the ANS are out of sync with each other. This can be likened to driving a car with one foot on the gas pedal (the sympathetic nervous system) and the other on the brake (the parasympathetic nervous system) at the same time – this creates a jerky ride, burns more gas, and isn’t great for your car, either! Likewise, the incoherent patterns of physiological activity associated with stressful emotions can cause our body to operate inefficiently, deplete our energy, and produce extra wear and tear on our whole system. This is especially true if stress and negative emotions are prolonged or experienced often.
In contrast, positive emotions send a very different signal throughout our body. When we experience uplifting emotions such as appreciation, joy, care, and love; our heart rhythm pattern becomes highly ordered, looking like a smooth, harmonious wave . This is called a coherent heart rhythm pattern. When we are generating a coherent heart rhythm, the activity in the two branches of the ANS is synchronized and the body’s systems operate with increased efficiency and harmony. It’s no wonder that positive emotions feel so good – they actually help our body’s systems synchronize and work better.
Heart rhythm patterns during different emotional states. These graphs show examples of real-time heart rate variability patterns (heart rhythms) recorded from individuals experiencing different emotions. The incoherent heart rhythm pattern shown in the top graph, characterized by its irregular, jagged waveform, is typical of stress and negative emotions such as anger, frustration, and anxiety. The bottom graph shows an example of the coherent heart rhythm pattern that is typically observed when an individual is experiencing a sustained positive emotion, such as appreciation, compassion, or love. The coherent pattern is characterized by its regular, sine-wave-like waveform. It is interesting to note that the overall amount of heart rate variability is actually the same in the two recordings shown above; however, the patterns of the HRV waveforms are clearly different.
The effect of heart activity on brain function has been researched extensively over about the past 40 years. Earlier research mainly examined the effects of heart activity occurring on a very short…
Source: When heart is under stress, our brain cannot think well
The effect of heart activity on brain function has been researched extensively over about the past 40 years. Earlier research mainly examined the effects of heart activity occurring on a very short time scale – over several consecutive heartbeats at maximum. Scientists at the Institute of HeartMath have extended this body of scientific research by looking at how larger-scale patterns of heart activity affect the brain’s functioning.
HeartMath research has demonstrated that different patterns of heart activity (which accompany different emotional states) have distinct effects on cognitive and emotional function. During stress and negative emotions, when the heart rhythm pattern is erratic and disordered, the corresponding pattern of neural signals traveling from the heart to the brain inhibits higher cognitive functions. This limits our ability to think clearly, remember, learn, reason, and make effective decisions. (This helps explain why we may often act impulsively and unwisely when we’re under stress.) The heart’s input to the brain during stressful or negative emotions also has a profound effect on the brain’s emotional processes—actually serving to reinforce the emotional experience of stress.
In contrast, the more ordered and stable pattern of the heart’s input to the brain during positive emotional states has the opposite effect – it facilitates cognitive function and reinforces positive feelings and emotional stability. This means that learning to generate increased heart rhythm coherence, by sustaining positive emotions, not only benefits the entire body, but also profoundly affects how we perceive, think, feel, and perform.
When it comes to naturally boosting the immune system by optimizing its response to diseases and infections, beta glucans are crucial weapons in the fight to stay healthy. But because the body doesn’t produce beta glucans naturally, the only way to get the compound is through outside sources—namely, baker’s yeast, shiitake mushrooms, and cereal grains, like barley, oats, rye, and wheat. After a century and a half of research,1 studies have shown that beta glucans act as immunomodulator agents, meaning they trigger a cascade of events that help regulate the immune system, making it more efficient. Specifically, beta glucans stimulate the activity of macrophages, which are versatile immune cells that ingest and demolish invading pathogens and stimulate other immune cells to attack.2 Macrophages also release cytokines, chemicals that when secreted enable the immune cells to communicate with one another. In addition, beta glucans stimulate lethal white blood cells (lymphocytes) that bind to tumors or viruses, and release chemicals to destroy it. Beta Glucans and Heart HealthNo doubt you’ve heard about the heart health benefits associated with a diet that includes consuming oats. That’s partly because oats are good sources of the soluble fiber beta glucan. In a study conducted by the US Department of Agriculture’s Beltsville Human Nutrition Research Center in Maryland, beta glucan was concentrated into an oat fiber extract so it could be easily incorporated into a typical diet. Male and female study participants with mildly high cholesterol were put on a maintenance diet for one week and then were given an oat fiber extract containing either 1% or 10% beta glucan. After five weeks of receiving the beta glucan extract, both groups showed a significant reduction of total cholesterol and low density lipoprotein (LDL).What’s more, total cholesterol levels were significantly lower in the group who received the higher beta glucan extract diet than in those on the low beta glucan diet.3 Mounting Support to Fight CancerWith about 560,000 deaths from cancer each year,4 researchers have spent decades searching for substances with cancer-fighting properties. Lentinan, a type of beta glucan found in shiitake mushrooms, is believed to reduce tumor activity and lessen the side effects of cancer treatment.5 Researchers at Teikyo University’s Biotechnology Research Center in Kawasaki, Japan, showed that lentinan has anti-tumor properties, suppressing the formation and development of tumors.
“Results of the clinical application of lentinan have proven prolongation of life span of the patients with advanced and recurrent stomach, colorectal, and breast cancer with only little toxic side effect,” wrote the study’s authors. It also appears that lentinan restores or boosts the responsiveness of cytokines, which interact with immune cells and regulate the response to the disease.6 In an earlier Japanese study, mice with tumors that received beta glucans, including lentinan, experienced a rapid decrease in the number of tumor cells as well as a notable increase in neutrophils in solid tumors.7 Neutrophils are a type of white blood cell that destroys invaders—in this case, cancerous cells—by ingesting them and using chemicals to break them down. In fact, beta glucans have been used as an immunoadjuvant therapy (an immune system stimulant) for cancer since 1980, mostly in Japan.1 Helping the Body Conquer InfectionsIn addition to beta glucan’s assistance in the fight against cancer, studies show they also help the body do battle with bacteria resistant to antibiotic treatment and viruses that cause upper respiratory infections. At Brigham and Women’s Hospital in Boston, Massachusetts, researchers found that the compound enhances antibiotic efficacy in rats infected with antibiotic-resistant bacteria. Rats with intra-abdominal sepsis due to antibiotic-resistant bacteria—namely, Escherichia coli or Staphylococcus aureus—were given a type of beta glucan (PGG glucan) that enhances the function of macrophages and neutrophils. Researchers looked at beta glucan’s ability to work in partnership with antibiotics to decrease mortality of the rats. “Results of these studies demonstrated that prophylaxis with PGG glucan in combination with antibiotics provided enhanced protection against lethal challenge with Escherichia coli or Staphylococcus aureus as compared with the use of antibiotics alone,” wrote the researchers.8 Further animal research highlights beta glucan’s impact on a form of Escherichia coli (ETEC), the culprit behind traveler’s diarrhea. Belgian scientists orally administered three different beta-glucans in pigs with an ETEC infection that had just been weaned. The study found that pigs fed for two weeks after weaning with the glucans were less susceptible to the infection (evidenced by a lower incidence of diarrhea) compared to the control group. “This study showed that beta-glucans can protect against an ETEC infection,” concluded the researchers. “To our knowledge, this is the first in vivo study, in which the use of beta-glucans as feed ingredient for just-weaned piglets was tested for their protective effects against ETEC infection.”9
Beta glucans also appear to mitigate the symptoms of the common cold—or at least reduce the number of days people call in sick to work. The Montana Center for Work Physiology and Exercise Metabolism examined beta glucans’ ability to mitigate upper respiratory infections in a single blind, randomized trial in 2008. The scientists chose firefighters as their subjects since they are regularly bombarded with smoke and fumes as they battle blazes and are more susceptible to respiratory troubles as a result. Researchers provided the group of firefighters with either a beta-glucan-containing supplement or a placebo and asked participants to write down any cold symptoms (runny or stuffy nose, sore throat, coughing, sneezing, colored discharge) or flu symptoms (fever, headache, general aches and pains, fatigue and weakness, chest discomfort, cough). Firefighters who recorded having these symptoms for two consecutive days were classified as having an upper respiratory tract infection. Participants who consumed the supplement had fewer (23%) upper respiratory tract infections, compared to the group of firefighters taking a placebo. “The results are consistent with previous clinical research involving marathoners, individuals with high stress lifestyles and the general population,” wrote Brent C. Rudy, the director of the Montana Center for Work Physiology and Exercise Metabolism.10 SummaryAlthough there isn’t a single magic bullet when it comes to eliminating disease, scores of research shows that adding beta glucans to your daily diet—either in the form of supplements or foods that contain the compound—can play a significant role in helping your body fend off not only the common cold and respiratory infections but also more serious diseases, including cancer. “Beta glucans have been used as an immunoadjuvant therapy (an immune system stimulant) for cancer since 1980, mostly in Japan.” If you have any questions on the scientific content of this article, please call a Life Extension® Health Advisor at 1-866-864-3027. |
|||
| References | |||
| 1. J Immunotoxicol. 2008 Jan;5(1):47-57.
2. http://www.niaid.nih.gov/publications/immune/the_immune_system.pdf. 3. J Am Coll Nutr.1997 Feb;16(1):46-5. 5. http://www.cancer.org/docroot/ETO/content/ETO_5_3X_Shiitake_Mushroom.asp?sitearea=ETO. 6. Dev Biol Stand. 1992;77:191-7. 7. Hum Cell. 1990 Jun;3(2):124-30. 8. Ann N Y Acad Sci. 1996 Oct 25;797:285-7. 9. Vet Immunol Immunopathol. 2009 Mar 15;128(1-3):60-6. 10. http://www2.prnewswire.com/cgi-bin/stories.pl?ACCT=109&STORY=/www/story/05-29-2008/0004822407&EDATE=. |

Beta-glucans are a group of structurally similar plant compounds that deliver significant health benefits. Although a few of these indigestible substances qualify as insoluble fiber, most are soluble in nature. Compared to other types of soluble fiber, beta-glucans are especially effective at achieving and maintaining healthy cholesterol levels, which makes them particularly useful for protecting against heart disease. Whole grains, mushrooms and yeast products are among the richest sources of beta-glucans.
Beta-glucans are found in the bran of many common whole grains, with oats and barley containing the highest amounts. According to the “Encyclopedia of Healing Foods,” consuming 3 grams of soluble oat fiber each day – which is the amount supplied by a single serving of oatmeal or oat bran cereal – has been shown to reduce high cholesterol levels by as much as 23 percent. The cholesterol-reducing effects of the beta-glucans in these two grains are so well established that the U.S. Food and Drug Administration allows whole oat and barley products to carry the “heart healthy” label. Whole wheat and rye are lower in beta-glucan fiber than oats and barley, but still contain significant amounts.
Mushrooms – particularly shiitake, maitake, reishi, shimeji and oyster varieties – are also good sources of beta-glucans. In addition to promoting healthy cholesterol levels, the beta-glucans in mushrooms have been shown to boost immune system function. They do this by activating immune cell response and stimulating the production of white blood cells. According to the book “Superfoods: The Healthiest Foods on the Planet,” the beta-glucans in some mushrooms also help regulate immune system response to cancer, which may make them useful in controlling and treating the disease. The beta-glucans in maitake and shiitake mushrooms demonstrate particularly high levels of anti-carcinogenic activity.
A sweet, fiber-rich fruit native to the Middle East, dates are also high in beta-glucans. They contain beta-D-glucan, a special type that’s both soluble and insoluble in nature. Its solubility allows beta-D-glucan to bind to dietary cholesterol and remove it from your body through waste. Like the beta-glucans in whole grains and fungi, beta-D-glucan helps keep blood glucose levels even by slowing the rate at which the small intestine absorbs simple sugars. According to the “Encyclopedia for Healing Foods,” beta-D-glucan is also insoluble in nature and promotes bowel health and regularity by helping to produce larger, softer stools that are easy to eliminate.
Men and women through the age of 50 are generally advised to get 38 grams and 25 grams of total fiber a day, respectively. Since most older adults require fewer calories, men and women past the age of 50 have correspondingly lower fiber needs of 30 grams and 21 grams, respectively. Dietary guidelines suggest that consuming a wide range of fiber-rich foods and meeting daily intake recommendations increase your likelihood of getting sufficient amounts of soluble and insoluble fiber. Although beta-glucans are considered most effective at reducing high cholesterol, other types of soluble fiber – such as pectin – are also beneficial. Citrus fruits, apples, pears and dried beans, peas and lentils are all known to promote healthy cholesterol levels.
Beta-glucans are naturally occurring polysaccharides. These glucose polymers are constituents of the cell wall of certain pathogenic bacteria and fungi. The healing and immunostimulating properties of mushrooms have been known for thousands of years in the Eastern countries. These mushrooms contain biologically active polysaccharides that mostly belong to group of beta-glucans. These substances increase host immune defense by activating complement system, enhancing macrophages and natural killer cell function.
The induction of cellular responses by mushroom and other beta-glucans is likely to involve their specific interaction with several cell surface receptors, as complement receptor 3 (CR3; CD11b/CD18), lactosylceramide, selected scavenger receptors, and dectin-1 (betaGR). beta-Glucans also show anticarcinogenic activity.
They can prevent oncogenesis due to the protective effect against potent genotoxic carcinogens. As immunostimulating agent, which acts through the activation of macrophages and NK cell cytotoxicity, beta-glucan can inhibit tumor growth in promotion stage too. Anti-angiogenesis can be one of the pathways through which beta-glucans can reduce tumor proliferation, prevent tumor metastasis. beta-Glucan as adjuvant to cancer chemotherapy and radiotherapy demonstrated the positive role in the restoration of hematopiesis following by bone marrow injury. Immunotherapy using monoclonal antibodies is a novel strategy of cancer treatment. These antibodies activate complement system and opsonize tumor cells with iC3b fragment. In contrast to microorganisms, tumor cells, as well as other host cells, lack beta-glucan as a surface component and cannot trigger complement receptor 3-dependent cellular cytotoxicity and initiate tumor-killing activity. This mechanism could be induced in the presence of beta-glucans.
Over the last several years, research teams across the world have scoured the genomes of cancer cells in a quest to uncover the genes that drive tumor growth. Meticulous analyses of the many thousands of genetic mutations that can accumulate in each tumor’s DNA have brought an increasing number of “driver genes” into view. Now, scientists are also turning their attention to another key question: What are the biological processes that lead to these DNA mutations in the first place?
“The mutations we see in a patient’s tumor are an amalgamation — genetic relics left behind by multiple different processes in the cell,” says Gad Getz, director of Cancer Genome Computational Analysis at the Broad Institute and an associate professor at Massachusetts General Hospital (MGH). “We don’t know what those processes are up front, but with sophisticated computational methods, we can now uncover them, and in doing so, gain insights into the biology of different cancers and how they can be more effectively treated.”
In this week’s issue of Nature Genetics, Getz and his colleagues report the results of a deep analysis of nearly 300 urothelial tumors, a type of bladder cancer responsible for roughly 150,000 deaths per year worldwide. Their work highlights the role of a well-known DNA repair process, known as nucleotide-excision repair (NER) and, for the first time, reveals genomic evidence for the role of tobacco smoking in this form of cancer.
To retrace the biological steps that lead to genetic mutations in cancer, Getz and other leading cancer genomics experts are studying so-called “mutational signatures” — patterns of mutations that are left behind in tumor DNA and reflect different mutation-causing events or exposures. For example, cigarette smoke leaves a characteristic imprint on the genomes of lung cancer cells. This imprint (known as Signature 4 in the COSMIC database), can be uncovered by surveying the genomes of lung tumors through large-scale sequencing, and then harnessing advanced computational algorithms — pioneered by multiple groups, including Getz’s team — to reveal the precise arrangement of genetic typos that emerge from continued cigarette use. Similar approaches can reveal mutational signatures tied to other mutagens as well as sloppy housekeeping functions, like DNA maintenance and repair.
Although studies of driver genes rely on mutations that land in and around genes (and thus exert substantial biological effects), analyses of mutational signatures cast a wide view, examining mutations that land in any and all parts of the genome, including areas that carry little or no functional consequence.
“Here, we really aren’t concerned with the driver mutations,” says Getz. “Most of these mutations are passenger mutations that didn’t necessarily cause the cancer, but instead were byproducts of the biological forces that led to mutations in the tumor genome.”
Getz and his colleagues, including researchers working on The Cancer Genome Atlas (TCGA), recently uncovered some intriguing findings related to urothelial cancer. They found that the ERCC2 gene — a core component of the NER pathway — is frequently mutated in urothelial tumors. Importantly, this is the only tumor type found to date in which ERCC2 is significantly mutated beyond what is expected by chance. In addition, Broad and Dana-Farber researcher Eliezer Van Allen and his colleagues revealed an interesting association between ERCC2 mutations and cancer therapeutic response in bladder cancer: Patients carrying ERCC2 mutations were found to be more sensitive to cisplatin, a platinum-based chemotherapy that induces DNA damage, which is mended through NER as well as other repair pathways. Together, these findings suggested that DNA repair, specifically NER, might play an important role in bladder cancer.
To dig more deeply into this area, Getz’s team, led by first authors Jaegil Kim (Broad Institute), Ken Mouw (Dana-Farber), and Paz Polak (MGH) in collaboration with Dana-Farber’s Alan D’Andrea, began analyzing the mutational signatures — the characteristic patterns of DNA mutations — in a TCGA cohort of urothelial tumors. They identified four key signatures, including one that seems nearly identical to a previously uncovered signature that is shared across many tumor types, including bladder cancer. Despite its near ubiquity, the biological roots of this signature, known as Signature 5, are not known.
To probe it further, the researchers searched for mutated genes that are associated with increased Signature 5 activity in bladder cancer. Sure enough, their top find was ERCC2. This was true not only in the TCGA cohort, but also in two other independent collections of urothelial tumor samples.
“That was a big finding, because it suggests that the cause of the Signature 5 mutations might be related to DNA repair”, says Getz.
“We hypothesize that these mutations are generated across all cancers in the setting of increased levels of DNA damage from tobacco or other mutagens, but in most cancers, the DNA repair machinery works well and repairs most of them — so they are relatively infrequent.” adds Jaegil Kim, co-first author of the paper. Yet in bladder tumors that harbor ERCC2 mutations, the repair machinery is crippled, so Signature 5 DNA mutations persist and accrue over time.
Although ERCC2 was by far the strongest genetic association with Signature 5, Getz and his colleagues noticed that several tumors lacked the ERCC2 mutation yet nevertheless showed increased Signature 5 activity. Interestingly, a handful of these tumors carry mutations in a different NER pathway gene. Although the number is small (and not statistically significant), the discovery further underscores the role of this form of DNA repair in bladder cancer.
These results also add to scientists’ growing but far-from-complete knowledge of the mutational signatures linked with faulty DNA repair. So far, just two other modes of DNA repair (homologous recombination and mismatch-repair) have been assigned mutational signatures.
A smoking gun
For years, it has been known that smoking can raise a person’s risk of developing bladder cancer. Despite this clinical knowledge, there has been little additional proof to corroborate smoking’s biological impact in the disease.
Indeed, Getz and his colleagues did not see any of the typical “smoking” signatures in bladder tumor genomes. (These include Signature 4 as well as a related signature connected to chewing tobacco.) However, they did uncover a significant association with Signature 5 activity. Regardless of whether or not their tumors harbored ERCC2 mutations, patients who smoked showed a significantly higher prevalence of Signature 5 mutations than patients who did not.
“Prior to this work, there was no direct genomic evidence of how smoking contributes to mutations in bladder cancer, due to a lack of the typical smoking signatures,” says Kim. “Our study provides a genomic fingerprint to suggest its role biologically.”
In addition to reflecting the genomic impact of cigarette smoke in bladder cancer, Getz’s studies of Signature 5 provide other important clinical inroads. “Having a defect in a DNA repair mechanism could represent a vulnerability that can be targeted therapeutically,” says Getz.
Uncovering these vulnerabilities — not just in bladder cancer but also in other major tumor types — represents one of the holy grails of mutational signature studies. “As an international community, we are in the process of mapping these signatures across all cancers, explaining their biological meaning and hopefully uncovering new vulnerabilities that can be leveraged for more effective cancer treatments,” says Getz.
Papers cited
Kim J, Mouw KW, Polak P, et al. Somatic ERCC2 mutations are associated with a distinct genomic signature in urothelia tumors. Nature Genetics. Published online April 25, 2016.
Kasar S, Kim J, et al. Whole-genome sequencing reveals activation-induced cytidine deaminase signatures during indolent chronic lymphocytic leukaemia evolution. Nature Communications. 2015 6:8866
The Cancer Genome Atlas Research Network. Comprehensive molecular characterization of urothelial bladder carcinoma. Nature. 2014; 507:315-22.
Van Allen EM, et al. Somatic ERCC2 mutations correlate with cisplatin sensitivity in muscle-invasive urothelial carcinoma. Cancer Discovery. 2014; 4(10):1140-53.
Over the past few decades, the healthcare community has observed an intriguing phenomenon: diseases related to the immune system—type 1 diabetes (T1D) and other autoimmune diseases, allergies, and …
Source: Babies have better immune system in Karelia than in Finland
Copyright © 2026 KRM Digital Solutions