System that Could Reduce Neurodegeneration in Huntington’s Discovered
Summary: Researchers have identified a mechanism that may reduce the toxic aggregation of the huntingtin protein. The findings could lead to new treatment options for Huntington’s patients.
Source: University of Cologne.
Dr David Vilchez and his team at CECAD have made an important step towards understanding the mechanisms that cause the neurodegenerative disorder Huntington’s disease. Particularly, they identified a system blocking the accumulation of toxin protein aggregates, which are responsible for neurodegeneration. The results have now been published in the journal ‘Nature Communications’.
Huntington’s disease is a neurodegenerative disorder that results in the death of brain cells, leading to uncontrolled body movement, loss of speech and psychosis. Mutations in the huntingtin gene cause the disease, resulting in the toxic aggregation of the huntingtin protein. The accumulation of these aggregates causes neurodegeneration and usually leads to the patient’s death within twenty years after the onset of the disease.
To examine the mechanisms underlying Huntington’s disease, Vilchez and his team used so-called induced pluripotent stem cells (iPSC) from Huntington’s disease patients, which are able to differentiate into any cell type, such as neurons. Induced pluripotent stem cells derived from patients with Huntington’s disease exhibit a striking ability to avoid the accumulation of toxic protein aggregates, a hallmark of the disease. Even though iPSCs express the mutant gene responsible for Huntington’s disease, no aggregates were found.
The protein degradation process
Ubiquitination and targeting
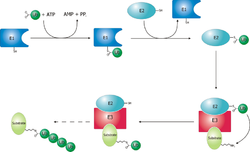
Proteins are targeted for degradation by the proteasome with covalent modification of a lysine residue that requires the coordinated reactions of three enzymes. In the first step, a ubiquitin-activating enzyme (known as E1) hydrolyzes ATP and adenylylates a ubiquitin molecule. This is then transferred to E1’s active-site cysteine residue in concert with the adenylylation of a second ubiquitin.[41] This adenylylated ubiquitin is then transferred to a cysteine of a second enzyme, ubiquitin-conjugating enzyme (E2). In the last step, a member of a highly diverse class of enzymes known as ubiquitin ligases (E3) recognizes the specific protein to be ubiquitinated and catalyzes the transfer of ubiquitin from E2 to this target protein. A target protein must be labeled with at least four ubiquitin monomers (in the form of a polyubiquitin chain) before it is recognized by the proteasome lid.[42] It is therefore the E3 that confers substrate specificity to this system.[43] The number of E1, E2, and E3 proteins expressed depends on the organism and cell type, but there are many different E3 enzymes present in humans, indicating that there is a huge number of targets for the ubiquitin proteasome system.
The mechanism by which a polyubiquitinated protein is targeted to the proteasome is not fully understood. Ubiquitin-receptor proteins have an N-terminal ubiquitin-like (UBL) domain and one or more ubiquitin-associated (UBA) domains. The UBL domains are recognized by the 19S proteasome caps and the UBA domains bind ubiquitin via three-helix bundles. These receptor proteins may escort polyubiquitinated proteins to the proteasome, though the specifics of this interaction and its regulation are unclear.[44]
The ubiquitin protein itself is 76 amino acids long and was named due to its ubiquitous nature, as it has a highly conserved sequence and is found in all known eukaryotic organisms.[45] The genes encoding ubiquitin in eukaryotes are arranged in tandem repeats, possibly due to the heavy transcription demands on these genes to produce enough ubiquitin for the cell. It has been proposed that ubiquitin is the slowest-evolving protein identified to date.[46] Ubiquitin contains seven lysine residues to which another ubiquitin can be ligated, resulting in different types of polyubiquitin chains.[47] Chains in which each additional ubiquitin is linked to lysine 48 of the previous ubiquitin have a role in proteasome targeting, while other types of chains may be involved in other processes.[48][49]
Unfolding and translocation
After a protein has been ubiquitinated, it is recognized by the 19S regulatory particle in an ATP-dependent binding step.[25] The substrate protein must then enter the interior of the 20S particle to come in contact with the proteolytic active sites. Because the 20S particle’s central channel is narrow and gated by the N-terminal tails of the α ring subunits, the substrates must be at least partially unfolded before they enter the core. The passage of the unfolded substrate into the core is called translocation and necessarily occurs after deubiquitination.[25] However, the order in which substrates are deubiquitinated and unfolded is not yet clear.[50] Which of these processes is the rate-limiting step in the overall proteolysis reaction depends on the specific substrate; for some proteins, the unfolding process is rate-limiting, while deubiquitination is the slowest step for other proteins.[24] The extent to which substrates must be unfolded before translocation is not known, but substantial tertiary structure, and in particular nonlocal interactions such as disulfide bonds, are sufficient to inhibit degradation.[51] The presence of intrinsically disordered protein segments of sufficient size, either at the protein terminus or internally, has also been proposed to facilitate efficient initiation of degradation.[52][53]
The gate formed by the α subunits prevents peptides longer than about four residues from entering the interior of the 20S particle. The ATP molecules bound before the initial recognition step are hydrolyzed before translocation. While energy is needed for substrate unfolding, it is not required for translocation.[24][25] The assembled 26S proteasome can degrade unfolded proteins in the presence of a non-hydrolyzable ATP analog, but cannot degrade folded proteins, indicating that energy from ATP hydrolysis is used for substrate unfolding.[24] Passage of the unfolded substrate through the opened gate occurs via facilitated diffusion if the 19S cap is in the ATP-bound state.[54]
The mechanism for unfolding of globular proteins is necessarily general, but somewhat dependent on the amino acid sequence. Long sequences of alternating glycine and alanine have been shown to inhibit substrate unfolding, decreasing the efficiency of proteasomal degradation; this results in the release of partially degraded byproducts, possibly due to the decoupling of the ATP hydrolysis and unfolding steps.[55] Such glycine-alanine repeats are also found in nature, for example in silk fibroin; in particular, certain Epstein–Barr virusgene products bearing this sequence can stall the proteasome, helping the virus propagate by preventing antigen presentation on the major histocompatibility complex.[56]

A cutaway view of the proteasome 20S core particle illustrating the locations of the active sites. The α subunits are represented as green spheres and the β subunits as protein backbones colored by individual polypeptide chain. The small pink spheres represent the location of the active-site threonine residue in each subunit. Light blue chemical structures are the inhibitor bortezomib bound to the active sites.
Proteolysis
The mechanism of proteolysis by the β subunits of the 20S core particle is through a threonine-dependent nucleophilic attack. This mechanism may depend on an associated water molecule for deprotonation of the reactive threonine hydroxyl. Degradation occurs within the central chamber formed by the association of the two β rings and normally does not release partially degraded products, instead reducing the substrate to short polypeptides typically 7–9 residues long, though they can range from 4 to 25 residues, depending on the organism and substrate. The biochemical mechanism that determines product length is not fully characterized.[57] Although the three catalytic β subunits have a common mechanism, they have slightly different substrate specificities, which are considered chymotrypsin-like, trypsin-like, and peptidyl-glutamyl peptide-hydrolyzing (PHGH)-like. These variations in specificity are the result of interatomic contacts with local residues near the active sites of each subunit. Each catalytic β subunit also possesses a conserved lysine residue required for proteolysis.[19]
Although the proteasome normally produces very short peptide fragments, in some cases these products are themselves biologically active and functional molecules. Certain transcription factors regulating the expression of specific genes, including one component of the mammalian complex NF-κB, are synthesized as inactive precursors whose ubiquitination and subsequent proteasomal degradation converts them to an active form. Such activity requires the proteasome to cleave the substrate protein internally, rather than processively degrading it from one terminus. It has been suggested that long loops on these proteins’ surfaces serve as the proteasomal substrates and enter the central cavity, while the majority of the protein remains outside.[58] Similar effects have been observed in yeast proteins; this mechanism of selective degradation is known as regulated ubiquitin/proteasome dependent processing (RUP).[59]
Ubiquitin-independent degradation
Although most proteasomal substrates must be ubiquitinated before being degraded, there are some exceptions to this general rule, especially when the proteasome plays a normal role in the post-translational processing of the protein. The proteasomal activation of NF-κB by processing p105 into p50 via internal proteolysis is one major example.[58] Some proteins that are hypothesized to be unstable due to intrinsically unstructured regions,[60] are degraded in a ubiquitin-independent manner. The most well-known example of a ubiquitin-independent proteasome substrate is the enzyme ornithine decarboxylase.[61] Ubiquitin-independent mechanisms targeting key cell cycle regulators such as p53 have also been reported, although p53 is also subject to ubiquitin-dependent degradation.[62] Finally, structurally abnormal, misfolded, or highly oxidized proteins are also subject to ubiquitin-independent and 19S-independent degradation under conditions of cellular stress.[63]
Evolution
The 20S proteasome is both ubiquitous and essential in eukaryotes. Some prokaryotes, including many archaea and the bacterial order Actinomycetales also share homologs of the 20S proteasome, whereas most bacteria possess heat shock genes hslV and hslU, whose gene products are a multimeric protease arranged in a two-layered ring and an ATPase.[64] The hslV protein has been hypothesized to resemble the likely ancestor of the 20S proteasome.[65] In general, HslV is not essential in bacteria, and not all bacteria possess it, whereas some protists possess both the 20S and the hslV systems.[64]Many bacteria also possess other homologs of the proteasome and an associated ATPase, most notably ClpP and ClpX. This redundancy explains why the HslUV system is not essential.
Sequence analysis suggests that the catalytic β subunits diverged earlier in evolution than the predominantly structural α subunits. In bacteria that express a 20S proteasome, the β subunits have high sequence identity to archaeal and eukaryotic β subunits, whereas the α sequence identity is much lower. The presence of 20S proteasomes in bacteria may result from lateral gene transfer, while the diversification of subunits among eukaryotes is ascribed to multiple gene duplication events.[64]
Cell cycle control
Cell cycle progression is controlled by ordered action of cyclin-dependent kinases (CDKs), activated by specific cyclins that demarcate phases of the cell cycle. Mitotic cyclins, which persist in the cell for only a few minutes, have one of the shortest life spans of all intracellular proteins.[1] After a CDK-cyclin complex has performed its function, the associated cyclin is polyubiquitinated and destroyed by the proteasome, which provides directionality for the cell cycle. In particular, exit from mitosis requires the proteasome-dependent dissociation of the regulatory component cyclin B from the mitosis promoting factor complex.[66] In vertebrate cells, “slippage” through the mitotic checkpoint leading to premature M phase exit can occur despite the delay of this exit by the spindle checkpoint.[67]
Earlier cell cycle checkpoints such as post-restriction point check between G1 phase and S phase similarly involve proteasomal degradation of cyclin A, whose ubiquitination is promoted by the anaphase promoting complex (APC), an E3 ubiquitin ligase.[68] The APC and the Skp1/Cul1/F-box protein complex (SCF complex) are the two key regulators of cyclin degradation and checkpoint control; the SCF itself is regulated by the APC via ubiquitination of the adaptor protein, Skp2, which prevents SCF activity before the G1-S transition.[69]
Individual components of the 19S particle have their own regulatory roles. Gankyrin, a recently identified oncoprotein, is one of the 19S subcomponents that also tightly binds the cyclin-dependent kinase CDK4 and plays a key role in recognizing ubiquitinated p53, via its affinity for the ubiquitin ligase MDM2. Gankyrin is anti-apoptotic and has been shown to be overexpressed in some tumor cell types such as hepatocellular carcinoma.[70]
Regulation of plant growth
In plants, signaling by auxins, or phytohormones that order the direction and tropism of plant growth, induces the targeting of a class of transcription factor repressors known as Aux/IAA proteins for proteasomal degradation. These proteins are ubiquitinated by SCFTIR1, or SCF in complex with the auxin receptor TIR1. Degradation of Aux/IAA proteins derepresses transcription factors in the auxin-response factor (ARF) family and induces ARF-directed gene expression.[71] The cellular consequences of ARF activation depend on the plant type and developmental stage, but are involved in directing growth in roots and leaf veins. The specific response to ARF derepression is thought to be mediated by specificity in the pairing of individual ARF and Aux/IAA proteins.[72]
Apoptosis
Both internal and external signals can lead to the induction of apoptosis, or programmed cell death. The resulting deconstruction of cellular components is primarily carried out by specialized proteases known as caspases, but the proteasome also plays important and diverse roles in the apoptotic process. The involvement of the proteasome in this process is indicated by both the increase in protein ubiquitination, and of E1, E2, and E3 enzymes that is observed well in advance of apoptosis.[73][74][75] During apoptosis, proteasomes localized to the nucleus have also been observed to translocate to outer membrane blebs characteristic of apoptosis.[76]
Proteasome inhibition has different effects on apoptosis induction in different cell types. In general, the proteasome is not required for apoptosis, although inhibiting it is pro-apoptotic in most cell types that have been studied. Apoptosis is mediated through disrupting the regulated degradation of pro-growth cell cycle proteins.[77] However, some cell lines — in particular, primary cultures of quiescent and differentiated cells such as thymocytes and neurons — are prevented from undergoing apoptosis on exposure to proteasome inhibitors. The mechanism for this effect is not clear, but is hypothesized to be specific to cells in quiescent states, or to result from the differential activity of the pro-apoptotic kinase JNK.[78] The ability of proteasome inhibitors to induce apoptosis in rapidly dividing cells has been exploited in several recently developed chemotherapy agents such as bortezomib and salinosporamide A.
Response to cellular stress
In response to cellular stresses – such as infection, heat shock, or oxidative damage – heat shock proteins that identify misfolded or unfolded proteins and target them for proteasomal degradation are expressed. Both Hsp27 and Hsp90—chaperone proteins have been implicated in increasing the activity of the ubiquitin-proteasome system, though they are not direct participants in the process.[79] Hsp70, on the other hand, binds exposed hydrophobic patches on the surface of misfolded proteins and recruits E3 ubiquitin ligases such as CHIP to tag the proteins for proteasomal degradation.[80] The CHIP protein (carboxyl terminus of Hsp70-interacting protein) is itself regulated via inhibition of interactions between the E3 enzyme CHIP and its E2 binding partner.[81]
Similar mechanisms exist to promote the degradation of oxidatively damaged proteins via the proteasome system. In particular, proteasomes localized to the nucleus are regulated by PARP and actively degrade inappropriately oxidized histones.[82] Oxidized proteins, which often form large amorphous aggregates in the cell, can be degraded directly by the 20S core particle without the 19S regulatory cap and do not require ATP hydrolysis or tagging with ubiquitin.[63] However, high levels of oxidative damage increases the degree of cross-linking between protein fragments, rendering the aggregates resistant to proteolysis. Larger numbers and sizes of such highly oxidized aggregates are associated with aging.[83]
Dysregulation of the ubiquitin proteasome system may contribute to several neural diseases. It may lead to brain tumors such as astrocytomas.[84] In some of the late-onset neurodegenerative diseases that share aggregation of misfolded proteins as a common feature, such as Parkinson’s disease and Alzheimer’s disease, large insoluble aggregates of misfolded proteins can form and then result in neurotoxicity, through mechanisms that are not yet well understood. Decreased proteasome activity has been suggested as a cause of aggregation and Lewy body formation in Parkinson’s.[85] This hypothesis is supported by the observation that yeast models of Parkinson’s are more susceptible to toxicity from α-synuclein, the major protein component of Lewy bodies, under conditions of low proteasome activity.[86] Impaired proteasomal activity may underlie cognitive disorders such as the autism spectrum disorders, and muscle and nerve diseases such as inclusion body myopathy.[84]
Role in the immune system
The proteasome plays a straightforward but critical role in the function of the adaptive immune system. Peptide antigens are displayed by the major histocompatibility complex class I (MHC) proteins on the surface of antigen-presenting cells. These peptides are products of proteasomal degradation of proteins originated by the invading pathogen. Although constitutively expressed proteasomes can participate in this process, a specialized complex composed of proteins, whose expression is induced by interferon gamma, are the primary producers of peptides which are optimal in size and composition for MHC binding. These proteins whose expression increases during the immune response include the 11S regulatory particle, whose main known biological role is regulating the production of MHC ligands, and specialized β subunits called β1i, β2i, and β5i with altered substrate specificity. The complex formed with the specialized β subunits is known as the immunoproteasome.[15] Another β5i variant subunit, β5t, is expressed in the thymus, leading to a thymus-specific “thymoproteasome” whose function is as yet unclear.[87]
The strength of MHC class I ligand binding is dependent on the composition of the ligand C-terminus, as peptides bind by hydrogen bonding and by close contacts with a region called the “B pocket” on the MHC surface. Many MHC class I alleles prefer hydrophobic C-terminal residues, and the immunoproteasome complex is more likely to generate hydrophobic C-termini.[88]
Due to its role in generating the activated form of NF-κB, an anti-apoptotic and pro-inflammatory regulator of cytokine expression, proteasomal activity has been linked to inflammatory and autoimmune diseases. Increased levels of proteasome activity correlate with disease activity and have been implicated in autoimmune diseases including systemic lupus erythematosus and rheumatoid arthritis.[15]
The proteasome is also involved in Intracellular antibody-mediated proteolysis of antibody-bound virions. In this neutralisation pathway, TRIM21 (a protein of the tripartite motif family) binds with immunoglobulin G to direct the virion to the proteasome where it is degraded.
Tip: Kill the virus in your brain with whole foods , adequate sleep and exercise in the sun.